
Animals are the gifts of nature to us!
However, they are facing a threatening crisis to their survival! We should be concerned as our co-existence is the only way forward into the future! Our lives literally depend on them!
What is the Issue?
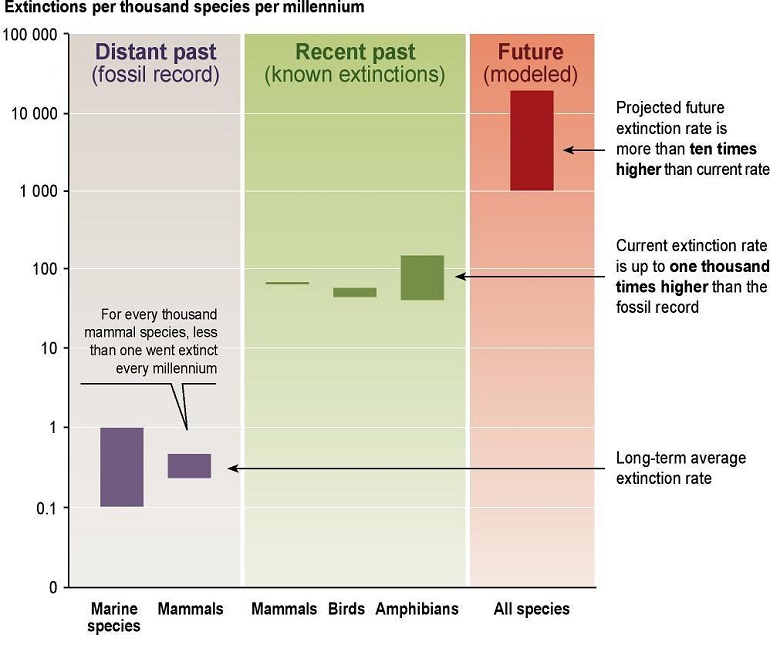
Today, the animal extinction rate is about 1,000 to 10,000 times faster than the natural rate because of human activities!
The world is witnessing a colossal decline in the wildlife population. Animals are suffering from loss of habitats, food shortage, climate change, over-exploitation, and human negligence!
Why Is It Important to Resolve This Problem?
Humans rely on animals for companionship, labor, fiber, and, most importantly, food. They play a significant role in the stability of the environment, ecosystem, and our lives.
Each animal, from the smallest fish to the largest land mammal plays a crucial role in the smooth running of our world. Animals help in pollination, keep the forest healthy and control pests.
Our future depends on them! Start making conscious choices to protect our wildlife! Say no to the ivory trade, report wildlife crimes, protect natural habitats, help reduce pollution, become a member of an NGO project, consume less and recycle more!
Different Animal Extinction Impacts
We exploit, hunt, and over-consume animals! We’re also devastating millions of habitats in our ever-growing greed for oil, gas, and timber.
Animal extinction severely affects the balance of ecosystems! All living things are interconnected through their interactions in a web of life.
A tiny change in nature’s distribution can cause debilitating effects on the entire world, including mankind!
What Can Be Done to Resolve This Problem?
We are friends of animals!
We are on a mission to raise awareness and outline all possible ways and solutions for the better conservation of animals. Join our hands to protect animals together!
How Do You End Neglected Animals?
You can start with animal rescue. You can also help reputable organizations to raise funds that aim to support animal welfare.
Wildlife, domesticated animals as well as pets – all are encountering the grim danger of extinction.
You can help these animals. Find out how:
1. Dogs
A man’s best friend! Dogs are a source of comfort and support! You’ll never feel lonely with a dog!
- Helping Dogs in Your Area
Help abandoned or lost dogs meet their owners or end up in a resounding animal shelter. That’s the easiest way to go about it!
- Adopt a Dog
Who doesn’t love a high-spirited and cuddly fur ball to play with at home? Life is definitely better with dogs. Adopt a rescue dog now!
- Spay or Neuter Your Dog
Be a responsible pet owner. Neutering decreases the chances of many diseases in your pets! Also helps in controlling populations!
- Teach Your Children to Value Animals Lives
Children tend to emulate their elders! Make a difference by leading a life worth following as an example!
Other Ways to Help
- Donate money, food and other essential!
- Donate to well-established and renowned animal shelters to help them continue doing their work.
- Volunteer your time at your local animal shelter or rescue!
Animal conservation includes their health and safety!
Take really good care of your pets! Feed them the best food available according to their needs!
It’s not hard to keep them secure these days with a plethora of dog collars and leashes available in the market! Always keep your pets on leashes when out and about in nature. Also, make sure your dog is safe and sound within the secure confines of your living space. The easiest way to keep an eye on your dog is with tracking dog collars, such as Halo and SpotOn; if you have not heard about these yet, read our Halo collar review and SpotOn fence reviews, or for a more economical option, check out Fi collar review.
2. Cats
Cute, fluffy, and adorable! Cats bring unconditional love and companionship to your life!
3. Fishes
Calm, serene, and beautiful creatures! Add beauty to our nature and bring peace to our ambiance!
4. Birds
An important member of our ecosystem! Crucial for people and the planet to thrive! Birds are great pollinators and also help to control pests.
Learn More About Endangered Species
According to the IUCN’s Red List of Threatened Species, over 26,500 species are in danger of extinction. These numbers are on the rise!
Humans are responsible for 99% of the endangered species on our planet. Learning about the predicament of animals is the first baby step taken in the right direction!


